For decades, cancer treatment meant one thing: chemotherapy. It attacked fast-growing cells - cancerous or not. Patients lost their hair, felt sick constantly, and often got sicker from the treatment than from the disease itself. But something has changed. Today, doctors don’t just look at where a tumor is - they look at what it’s made of. And that shift is saving lives.
What Targeted Therapy Really Means
Targeted therapy isn’t just another drug. It’s a completely different way of thinking about cancer. Instead of spraying poison across the whole body, it finds the exact genetic mistake that’s making the cancer grow. Think of it like a lock and key. Cancer cells have broken locks - mutated genes - that keep them multiplying out of control. Targeted drugs are the right key for those specific locks. They slip in, block the signal, and stop the cancer from growing - without wrecking healthy cells.The first big win came in 2001 with imatinib (Gleevec) for chronic myeloid leukemia. Before imatinib, only 20-30% of patients survived a year. After? Nearly 9 out of 10 did. That wasn’t just progress. It was a revolution.
How Tumor Genetics Guide Treatment
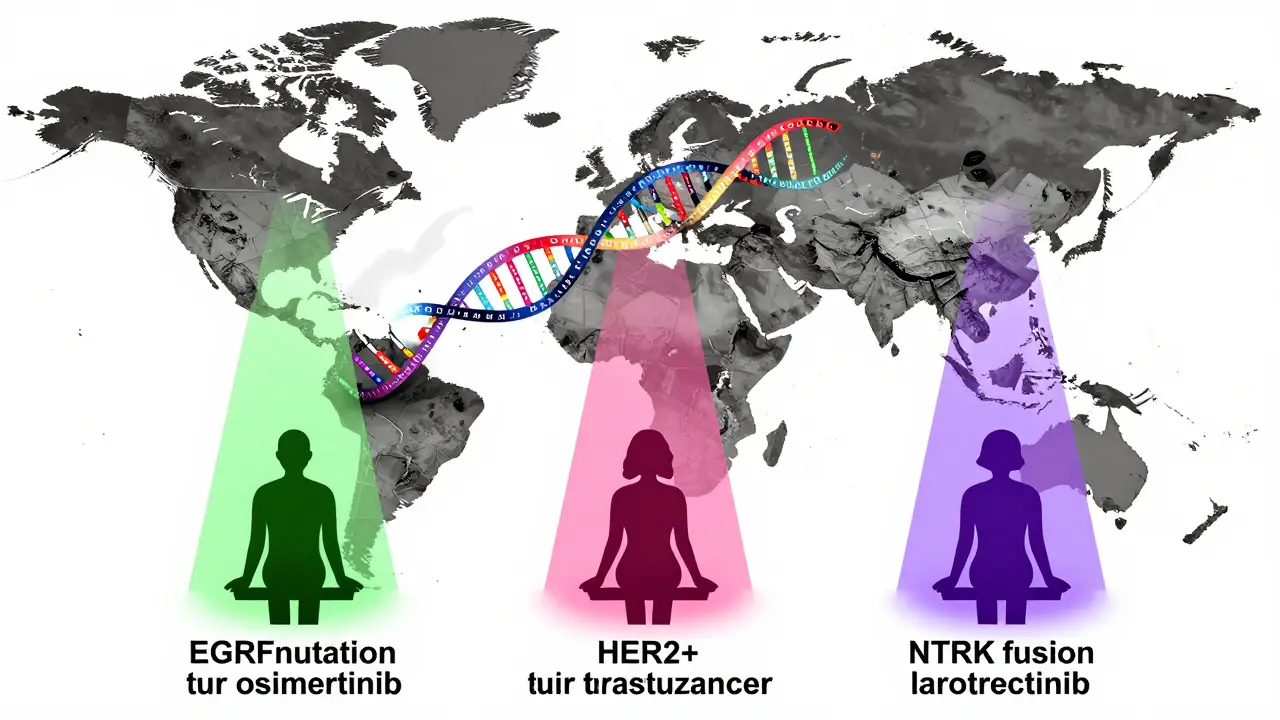
Every tumor has its own fingerprint. Two people with lung cancer might have completely different genetic drivers. One might have an EGFR mutation. Another might have an ALK fusion. A third might carry a RET change. Each needs a different drug.Here’s how it works in practice:
- EGFR mutations in non-small cell lung cancer? Osimertinib is the go-to. It cuts the risk of cancer spreading by more than half compared to old-school chemo.
- HER2-positive breast cancer? Trastuzumab locks onto the HER2 protein on the cell surface and tells the immune system to destroy it.
- NTRK fusions? Larotrectinib works across any cancer type - lung, thyroid, even rare pediatric tumors - if it has this specific genetic quirk. Response rates? 75%.
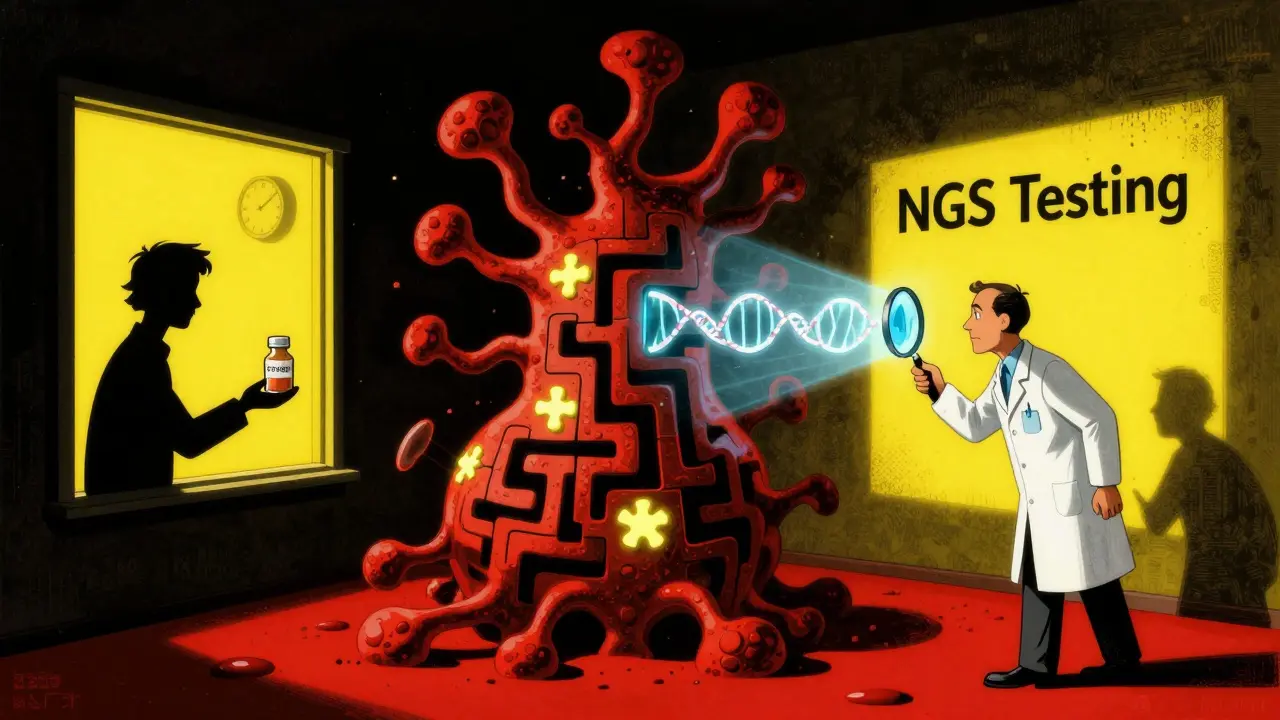
This is why testing matters. Before starting treatment, doctors now run a tumor through next-generation sequencing (NGS). Panels like FoundationOne CDx or MSK-IMPACT scan 300-500 cancer-related genes. It’s not cheap - around $5,500 - but it’s the only way to know which drug will actually work.
Why It Works Better Than Chemo - For Some
Let’s compare. A patient on traditional chemo might face nausea so bad they can’t eat, fatigue that leaves them in bed for days, or infections from a crashed immune system. Grade 3-4 side effects? Happen in 50-70% of cases.With targeted therapy? Those numbers drop to 15-30%. Patients often feel well enough to keep working, take care of kids, or travel. One patient with EGFR-mutant lung cancer posted on a cancer forum: “After starting osimertinib, my tumor shrank 80% in eight weeks. No vomiting. No hair loss. I went on a road trip.” That’s not a miracle. That’s science.
But here’s the catch: targeted therapy only works if the tumor has the right mutation. If you don’t test, you’re just guessing. And guessing means wasting time - and precious months - on drugs that won’t help.
The Hard Truth: Who Gets Left Behind
It’s tempting to think this is the future for everyone. But right now, only about 13.8% of cancer patients have a tumor with a currently targetable mutation. That’s according to AACR Project GENIE data from 2023. For many, there’s no drug yet.Even worse - access isn’t equal. In the U.S., 65% of advanced cancer patients get genomic testing. In Europe? Only 22%. In Asia? Just 8%. Insurance denials are common. One Reddit user wrote: “My NTRK fusion makes me eligible for larotrectinib. My insurance denied it because it’s ‘not standard for my cancer type.’” But the drug works regardless of where the tumor is. That’s the irony: science is ahead of policy.
Cost is another wall. A single month of targeted therapy can run $15,000 to $30,000. Chemo? $5,000-$10,000. A 2023 University of Chicago study found 40% of patients on targeted drugs faced serious financial hardship - debt, skipped meals, or delaying care.
The Resistance Problem
Targeted therapy isn’t a cure. Most patients develop resistance within 9 to 14 months. Why? Cancer is smart. It mutates again. The original lock changes shape. The key no longer fits.That’s why researchers are now testing combinations - pairing a targeted drug with something else, like an immunotherapy or a second targeted agent. Early trials show promise. For example, some lung cancer patients on osimertinib plus a new drug are seeing responses last over two years.
Liquid biopsies are helping too. These blood tests detect tumor DNA floating in the bloodstream. Guardant360, approved by the FDA in 2023, can spot resistance mutations months before a scan shows tumor growth. That means doctors can switch drugs before the cancer spreads.
Who’s Doing This Right?
Big academic centers like MD Anderson and Memorial Sloan Kettering have molecular tumor boards - teams of oncologists, pathologists, genetic counselors, and bioinformaticians who review every complex case. They’ve got the staff, the tech, and the time.But most community clinics? They don’t. Only 32% of community hospitals have these teams. And many oncologists aren’t trained to read genomic reports. ASCO recommends 40 hours of annual training on molecular oncology. How many are actually doing it?
That’s why groups like the Personalized Oncology Alliance step in. They offer free expert reviews to community practices - helping doctors who don’t have the resources to decode complex genetic reports.
What’s Next?
The next frontier? Targeting tumor suppressor genes. These are the brakes that cancer turns off - like TP53 or PTEN. They’re mutated in 80% of cancers. But so far, we have no drugs to fix them. That’s why experts say we’ve only scratched the surface.AI is helping. IBM Watson for Oncology matched molecular tumor board decisions 93% of the time in a 2024 study. That’s not to replace doctors - it’s to support them, especially in places with fewer experts.
And the FDA is adapting. In 2018, they approved pembrolizumab for any solid tumor with high microsatellite instability (MSI-H) - no matter where it was in the body. That was the first “tissue-agnostic” approval. More are coming.
By 2030, McKinsey predicts 40% of cancer patients will get biomarker-driven treatment. But if we don’t fix access, equity, and cost, that future will only belong to the lucky few.
What cancers are most likely to benefit from targeted therapy?
Lung cancer (especially non-small cell with EGFR, ALK, ROS1, or RET mutations), melanoma (BRAF mutations), breast cancer (HER2-positive), and certain blood cancers like chronic myeloid leukemia (BCR-ABL fusion) are the most common. But targeted therapies are now approved for over 42 cancer types - including rare ones with NTRK fusions or MSI-H status.
Do I need genetic testing even if my cancer is advanced?
Yes. For advanced cancers, molecular profiling is now considered standard of care by major oncology groups. Even if you’ve had chemo before, your tumor may have developed new mutations that make it eligible for a targeted drug. Testing can open doors to treatments you didn’t know existed.
Why is targeted therapy so expensive?
These drugs are complex to develop. They require years of research, specialized manufacturing, and extensive clinical trials focused on small patient groups. Companies recover R&D costs through high prices. But the cost is unsustainable - and it’s why insurance battles and patient hardship are so common.
Can targeted therapy cure cancer?
In rare cases - like early-stage CML with imatinib - patients can live decades without progression. For most, though, targeted therapy controls cancer as a chronic disease rather than curing it. The goal is to keep it stable, reduce symptoms, and extend life with good quality.
What if my tumor doesn’t have a known target?
You’re not out of options. Clinical trials are testing new drugs for rare mutations. Some programs, like NCI-MATCH, match patients to experimental therapies based on genetics, not tumor location. Ask your oncologist about trials. Even if you don’t qualify now, new targets are discovered every year.




9 Comments
Bridgette Pulliam-10 March 2026
I've seen this shift firsthand. My sister had stage IV lung cancer with an EGFR mutation. Chemo left her bedridden for weeks. Osimertinib? She started hiking again. No hair loss, no nausea. Just... living. It's not magic. It's biology finally catching up to the patient.
Mike Winter-11 March 2026
It's fascinating, isn't it? The precision of it all. We've moved from treating cancer as a monolithic enemy to recognizing it as a constellation of molecular anomalies. Each mutation, a unique signature. And yet... we're still playing whack-a-mole with resistance. The real revolution won't come until we predict evolution, not just react to it.
Randall Walker-12 March 2026
So... we spent decades poisoning people... and now we're giving them $20k/month pills? Cool. Glad we're 'progressing.'
Miranda Varn-Harper-14 March 2026
The data is clear. Targeted therapies are not panaceas. They are expensive, inaccessible, and often temporary. To frame them as a 'revolution' ignores the structural failures of healthcare systems. This isn't science advancing-it's capitalism repackaging hope as a subscription service.
Alexander Erb-14 March 2026
Honestly? This gives me chills. 😊 I work in a clinic that just got its first NGS machine last year. Seeing patients get answers after years of guessing? Pure gold. Even if it's not perfect, it’s lightyears ahead of where we were. And yeah, the cost sucks-but imagine if we didn’t try. 🙏
Donnie DeMarco-14 March 2026
Dude. I had a cousin on larotrectinib. Like, tumor just... vanished. Like a magic eraser on a whiteboard. And the best part? She didn’t look like a zombie. No puffy face, no zombie walk. Just her. Still drinking coffee. Still laughing. That’s the dream right there.
Tom Bolt-16 March 2026
Let me be clear: the fact that we’re still debating whether a patient deserves access to a life-saving drug based on zip code isn’t a failure of medicine-it’s a moral collapse. This isn’t about science anymore. It’s about who we choose to save. And right now? We’re choosing the wealthy.
Shourya Tanay-16 March 2026
From a translational oncology standpoint, the real bottleneck lies in the disconnect between genomic data acquisition and clinical decision-making infrastructure. The heterogeneity of mutational landscapes demands dynamic, adaptive frameworks-yet most oncology workflows remain anchored in 20th-century paradigms. The integration of AI-driven decision support systems, particularly those trained on multimodal datasets, represents the most viable pathway toward scalable precision oncology.
LiV Beau-16 March 2026
I just want to say-this is why I became a nurse. 💖 Seeing someone go from terrified to thriving because of a genetic test? That’s the kind of moment that keeps you going. Even if the system’s broken, the science? It’s beautiful. And we’re not done yet. Keep pushing. Keep testing. Keep fighting. 🙌