If you've ever stood at a pharmacy counter and been asked if you'd like the generic version of your prescription, you probably assumed that all generics are created equal. In reality, there is a hidden divide in the pharmaceutical world. Not all "generics" are the same. While both aim to lower your bill, there is a massive difference between an authorized generic and a traditional one. One is essentially the brand-name drug in a plain wrapper, while the other is a copycat created by a different company.
| Feature | Authorized Generic | Traditional Generic |
|---|---|---|
| Ingredients | Identical to brand (Active & Inactive) | Same active; different inactive allowed |
| Manufacturer | The original brand company | A separate generic company |
| FDA Pathway | NDA (No new approval needed) | ANDA (Requires bioequivalence proof) |
| Market Entry | Can enter before patent expires | Usually enters after patent expires |
| Orange Book | Not listed | Listed for therapeutic equivalence |
What exactly is an authorized generic?
Think of an Authorized Generic is an approved brand-name drug that is marketed without the brand name on its label. Essentially, the company that invented the drug decides to sell the exact same product under a generic name. There is no change in the formula, the chemical makeup, or the manufacturing process. The only thing that changes is the packaging and the price tag.
Because the FDA (Food and Drug Administration) had already approved the original drug through a New Drug Application (NDA), the manufacturer doesn't need to go through a new approval process to launch an authorized generic. They just notify the regulators that they are selling it without the brand name. This makes the rollout incredibly fast compared to other generics.
How do traditional generics work?
A Traditional Generic is a different beast. These are developed by other pharmaceutical companies that want to create a bioequivalent version of a brand-name drug. To do this, they must submit an Abbreviated New Drug Application (ANDA).
The "abbreviated" part is key. These companies don't have to repeat the expensive clinical trials to prove the drug is safe and effective-the FDA already knows it is because of the original brand. Instead, the generic maker only has to prove bioequivalence. This means they must show that the drug reaches the same part of the body, at the same speed and in the same amount as the original.
Here is the catch: while the active ingredient must be the same, the inactive ingredients (fillers, binders, or dyes) can differ. For most people, this doesn't matter. However, for some patients, these small changes in the "inactive" recipe can affect how they respond to the medication.
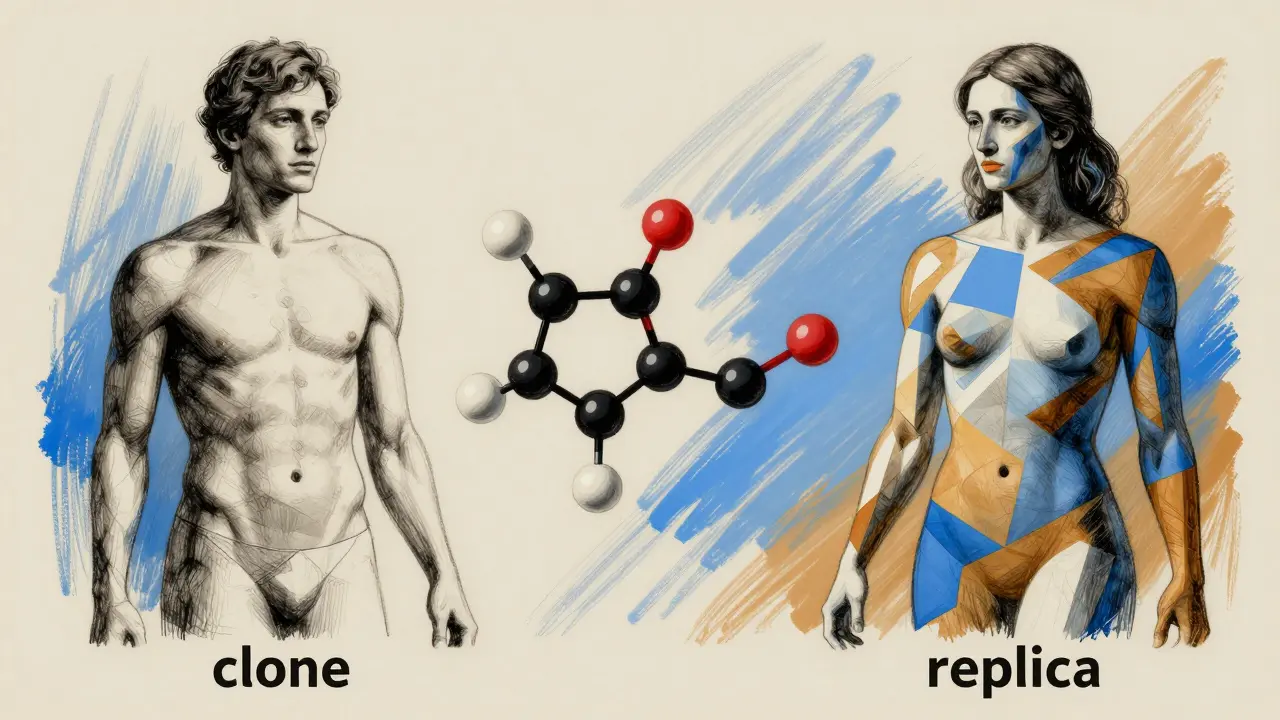
The core differences you should know
The main tension between these two types of drugs comes down to composition and timing. Authorized generics are clones; traditional generics are replicas.
- Composition: An authorized generic is a mirror image of the brand. It uses the same inactive ingredients. A traditional generic is a chemical match for the active part but may use different fillers.
- The Orange Book: Most pharmacists use the Orange Book (the FDA's official list) to see if a generic is therapeutically equivalent to a brand. Interestingly, authorized generics aren't listed here because they don't need separate approval-they *are* the brand drug.
- Timing: Traditional generics usually have to wait for a patent to expire. Authorized generics can actually enter the market *before* a patent expires because the patent holder is the one selling them.
Why does this matter for your health?
For the vast majority of medications, like a standard blood pressure pill, the difference is negligible. You'll get the same result regardless of which generic you take. But for certain "narrow therapeutic index" drugs-where a tiny change in dose or absorption can lead to a big change in effect-the distinction is vital.
Take asthma medications as an example. Some patients find that the specific formulation of a brand-name inhaler works better for them. In this case, an authorized generic is a lifesaver because it provides that exact same formulation at a lower cost. Traditional generics in the respiratory space can sometimes vary slightly in how they deliver the drug, which can be frustrating for sensitive patients.
If you have a history of reacting poorly to different generic brands, you might want to ask your pharmacist if an authorized generic is available. It removes the guesswork regarding inactive ingredients.

The strategic game: Why brands do this
You might wonder why a company would sell a cheaper, unbranded version of their own expensive drug. It's all about market share. When a patent is about to expire, a wave of traditional generics usually floods the market, crashing the price. By launching an authorized generic, the brand company can:
- Capture the price-sensitive customers who would have switched to a competitor.
- Keep a foothold in the generic market while still selling the high-priced brand version to others.
- Control the quality and supply chain of the "generic" alternative.
Essentially, it's a defensive move. Instead of letting a third-party company take all the generic profits, the original manufacturer takes a slice for themselves.
Navigating your prescription
If you are trying to save money but are worried about switching from a brand-name drug, here is a practical way to handle it. First, check if your insurance has a preferred generic on their formulary. If they do, it might be a traditional generic.
If you prefer the brand but can't afford it, ask your doctor to write "Dispense as Written" (DAW) or "Do Not Substitute" on the prescription. This forces the pharmacy to provide the brand. However, if you find an authorized generic, you get the best of both worlds: the brand's exact formula and the generic's price.
Are authorized generics safer than traditional generics?
Not necessarily "safer," but they are more consistent. Since they are identical to the brand-name drug, there is zero risk of a different inactive ingredient causing a reaction. Both are FDA-approved and held to the same strict quality standards.
Why aren't authorized generics listed in the Orange Book?
The Orange Book tracks drugs that have gone through the ANDA process to prove they are equivalent to a brand. Because an authorized generic *is* the brand drug, it doesn't need to prove equivalence-it already is the reference product.
Will my insurance cover an authorized generic?
Most insurance companies will cover them because they are typically cheaper than the brand-name version. However, coverage depends on your specific plan's formulary. It's always best to check with your provider.
Can I tell the difference between the two just by looking at the pill?
Usually, no. While they might have different colors or markings, you cannot identify whether a pill is an authorized or traditional generic without checking the manufacturer's information provided by the pharmacist.
Do authorized generics cost the same as traditional generics?
They are both significantly cheaper than brand-name drugs, but their prices can vary. Sometimes authorized generics are slightly more expensive than traditional ones because they are produced by the original brand manufacturer.



Write a comment