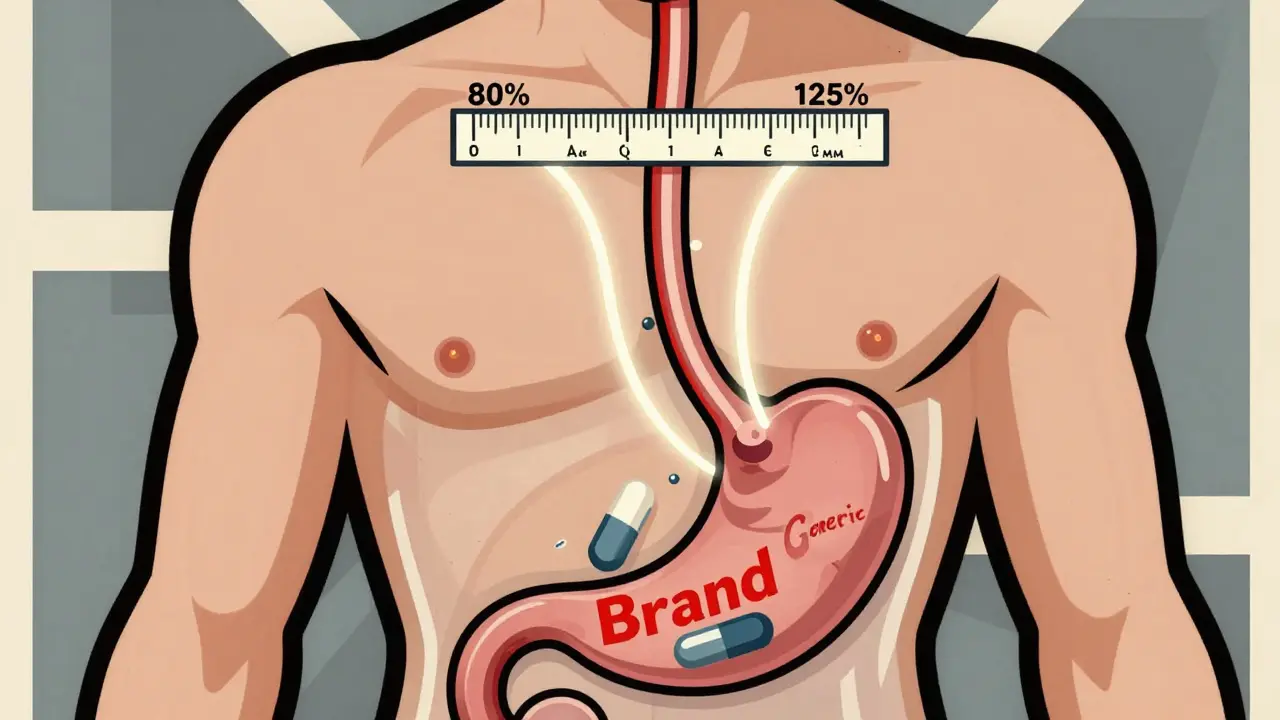
When you pick up a generic version of your prescription, you might assume it’s just a cheaper copy. But here’s the real question: is it really the same? The answer lies in something most people never hear about-the 80-125% rule. This isn’t about how much active ingredient is in the pill. It’s about how your body absorbs it. And it’s the reason millions of people safely use generic drugs every day.
What the 80-125% Rule Actually Means
The 80-125% rule is a global standard used by the FDA, EMA, WHO, and other health agencies to decide if a generic drug works like its brand-name version. It doesn’t say the generic has to contain 80% to 125% of the active ingredient. That’s a common myth. Instead, it looks at how your body handles the drug after you take it.
Researchers measure two key things: AUC (Area Under the Curve) and Cmax (maximum concentration). AUC tells you how much of the drug your body absorbs over time. Cmax tells you how fast it gets into your bloodstream. These aren’t guesses-they’re measured in clinical studies using blood samples from healthy volunteers.
The rule says the 90% confidence interval of the ratio between the generic and brand-name drug must fall entirely between 80% and 125%. That means if the brand drug gives you 100 units of exposure, the generic must deliver between 80 and 125 units. Not 79, not 126. The whole interval must fit inside that range.
Why 90%? Because it allows for a 5% error on each side, totaling 10%. This is stricter than it sounds. If the confidence interval even brushes outside 80% or 125%, the drug fails. No exceptions.
Why Logarithms? The Hidden Math Behind the Rule
Here’s where it gets technical-and important. Pharmacokinetic data like AUC and Cmax don’t follow a normal bell curve. They follow a log-normal distribution. That means the differences aren’t linear. A 20% increase isn’t the same as a 20% decrease in how your body responds.
So scientists transform the data using logarithms. On this scale, 80% becomes -0.2231 and 125% becomes +0.2231. This makes the range symmetrical and mathematically sound. The 80-125% rule only works because of this transformation. Skip this step, and the whole analysis is wrong.
Most people don’t need to do the math. But they should know: this isn’t arbitrary. It’s based on decades of data and statistical modeling. The FDA adopted it in the 1980s after reviewing hundreds of studies. Experts concluded that differences under 20% in absorption were unlikely to affect patient outcomes.
It’s Not Just About the Pill-It’s About Your Body
A lot of confusion comes from thinking the rule is about the amount of drug in the tablet. It’s not. Both brand and generic pills must contain 95% to 105% of the labeled active ingredient. That’s a standard manufacturing requirement. The 80-125% rule looks at what happens after you swallow it.
Imagine two identical pills. One is brand, one is generic. Same amount of drug. But the generic dissolves slower. Or the coating changes how it’s absorbed. Or your gut processes it differently. These small differences matter. The 80-125% rule catches them.
For example, a generic version of a blood thinner like warfarin might dissolve 10% slower. That could mean the peak concentration hits later. In most cases, it’s harmless. But for drugs with a narrow therapeutic index-where the difference between effective and toxic is razor-thin-those small delays matter. That’s why some drugs, like levothyroxine, require tighter limits: 90-111% instead of 80-125%.

What Happens When a Drug Fails the Test?
Not every generic passes. If the 90% confidence interval falls outside 80-125%, the FDA rejects it. No second chances. That’s why some generic drugs take years to develop. Manufacturers might run 3-4 different formulations before one works.
Highly variable drugs are especially tricky. If a drug’s absorption varies a lot between people (with a within-subject coefficient of variation over 30%), the standard rule doesn’t work. That’s why regulators use scaled average bioequivalence (SABE). This method adjusts the acceptance range based on how variable the reference drug is. For example, a drug with high variability might have a range of 69.84-143.19% for Cmax. That sounds wide-but it’s mathematically justified.
Complex formulations like inhalers, patches, or extended-release tablets are even harder. The FDA’s Complex Generics Initiative, launched in 2018, spends $35 million a year just to figure out how to test these properly. Some products still can’t be approved using the standard 80-125% rule.
Real-World Evidence: Do Generics Actually Work?
Despite the myths, the data is clear. A 2020 FDA analysis of over 2,000 generic drugs approved between 2003 and 2016 found that only 0.34% needed label changes due to bioequivalence issues after they hit the market. That’s less than 1 in 300.
Another study looked at 214 bioequivalence studies across 37 drug classes. All products that met the 80-125% rule showed no clinically meaningful differences in outcomes. Not in blood pressure control. Not in seizure frequency. Not in cholesterol reduction.
And yet, misinformation persists. A 2022 survey found 63% of community pharmacists thought the rule meant generics could contain 80-125% of the active ingredient. That’s not just wrong-it’s dangerous. It fuels patient fear. Reddit threads, pharmacy forums, and patient blogs are full of people worried their generic is “only 80% as strong.”
But here’s the truth: if your generic passed the bioequivalence test, it’s as effective as the brand. The difference isn’t in the pill. It’s in your perception.

Where the Rule Falls Short
The 80-125% rule isn’t perfect. It was never meant to be a universal solution. It was designed as a practical, one-size-fits-all standard to speed up generic approvals. Experts at the 1986 FDA Bioequivalence Hearing warned that the range might not suit every drug. That’s still true.
Anti-epileptic drugs are a major concern. A 2022 survey of 412 neurologists found 28% reported occasional problems with generic substitutions. Only 4% blamed bioequivalence standards-but the rest cited formulation differences, fillers, or coating changes. These aren’t covered by the 80-125% rule. They’re about how the pill is made, not how it’s absorbed.
Also, the rule doesn’t account for genetics. Someone with a slow-metabolizing liver enzyme might respond differently to a generic than someone with a fast one. That’s the next frontier. Research into pharmacogenomics suggests future bioequivalence standards might need to be personalized.
For now, the rule works. It’s not perfect, but it’s the best tool we have. And it’s saved billions in healthcare costs.
Why This Matters for You
Generic drugs make up 90% of prescriptions in the U.S. but only 23% of drug spending. Without the 80-125% rule, those savings wouldn’t exist. Every time you choose a generic, you’re trusting this system. And it’s been tested-not just in labs, but in real patients over decades.
If you’re on a critical medication like warfarin, levothyroxine, or an anti-seizure drug, talk to your doctor before switching. But if you’re on a statin, an antibiotic, or a blood pressure pill, the evidence is overwhelming: your generic is safe and effective.
The 80-125% rule isn’t magic. It’s science. And it’s working.
Does the 80-125% rule mean generic drugs contain 80% to 125% of the active ingredient?
No. This is a common misunderstanding. The 80-125% rule applies to the ratio of pharmacokinetic parameters-specifically AUC and Cmax-measured in clinical studies. It reflects how much of the drug enters your bloodstream and how fast. All generic drugs must contain 95% to 105% of the labeled active ingredient, just like brand-name drugs. The rule is about absorption, not content.
Why is a 90% confidence interval used instead of a 95% one?
The 90% confidence interval is used because it allows for a 5% error on each side (upper and lower), totaling 10% overall. This balances statistical rigor with practicality. In bioequivalence, the goal isn’t to prove the drugs are identical-it’s to prove they’re similar enough to be therapeutically interchangeable. A 95% interval would be too strict and make it harder to approve generics that are clinically equivalent.
Are there exceptions to the 80-125% rule?
Yes. For drugs with a narrow therapeutic index-like warfarin, levothyroxine, or phenytoin-tighter limits of 90-111% are required. For highly variable drugs, regulators use scaled average bioequivalence (SABE), which can expand the range up to 69.84-143.19% for Cmax. These exceptions are based on the drug’s behavior in the body, not arbitrary rules.
Do all countries use the same 80-125% rule?
Yes. The U.S. FDA, European Medicines Agency (EMA), World Health Organization (WHO), Health Canada, and China’s NMPA all use the same standard: the 90% confidence interval of the geometric mean ratio for AUC and Cmax must fall within 80-125%. This global harmonization allows generic manufacturers to develop one product for multiple markets.
Can a generic drug fail bioequivalence testing even if it has the same active ingredient?
Absolutely. Two drugs can have identical active ingredients but differ in inactive ingredients, coating, particle size, or dissolution rate. These differences can change how quickly or completely the drug is absorbed. If the 90% confidence interval for AUC or Cmax falls outside 80-125%, the generic is rejected-even if it contains 100% of the labeled drug.


14 Comments
bill cook- 1 March 2026
So let me get this straight - you’re telling me some guy in a lab in India made a pill that’s technically 80% as strong as my brand-name drug and I’m supposed to just swallow it like it’s fine? My blood pressure isn’t a fucking math problem. I’ve seen what happens when people switch generics. I know people. Real people. Not data points. And yeah, some of them crashed. Hard. This rule is a corporate loophole dressed up as science.
Katherine Farmer- 2 March 2026
There’s a fundamental misunderstanding here that permeates public discourse. The 80–125% rule is not a threshold of efficacy - it’s a statistical boundary for geometric mean ratios of AUC and Cmax under log-normal transformation. The FDA adopted it because it aligns with the concept of therapeutic equivalence, not chemical identity. To conflate bioavailability with active ingredient concentration is to confuse pharmacokinetics with pharmacy compounding. This is why laypeople panic when they see ‘80%’ - they think it means potency, not exposure. The rule is elegant. The public perception? Not so much.
Angel Wolfe- 3 March 2026
They’re lying. All of them. The FDA, WHO, the whole global pharma cartel. Why do you think they push generics so hard? To control you. To make you dependent on cheap pills that don’t work right. My cousin took a generic for his heart med and his kidneys started failing. They didn’t even test it on people like him - just healthy college kids. And now they say it’s ‘bioequivalent’? Bullshit. They’re cutting corners so Big Pharma can keep charging $500 for the brand. You think they care if you die? Nah. They care about quarterly profits. This rule? It’s a cover-up.
Vikas Meshram- 4 March 2026
Actually the 80-125% rule is based on the 90% confidence interval of the geometric mean ratio of AUC and Cmax which is derived from log-transformed data because pharmacokinetic parameters are log-normally distributed. The 20% range is not arbitrary but based on empirical data from hundreds of studies showing that differences within this range rarely affect clinical outcomes. Also the 95-105% active ingredient requirement is separate and enforced by cGMP. So no, generics are not '80% strong' - that's a myth perpetuated by misinformation. The science is solid.
Ben Estella- 4 March 2026
Look I don’t care about your fancy stats. I’ve been on the same generic for five years and I’m fine. My bloodwork’s perfect. My doctor says it’s the same. Why are we even having this conversation? The system works. People who say it doesn’t are either paranoid or got scammed by some scammy pharmacy. We’re not in Europe where they overthink everything. Here in America we trust the science. And the science says generics save lives and money. End of story.
Jimmy Quilty- 5 March 2026
Did you know that the 80-125% rule was originally proposed by a guy who worked for Pfizer back in the 80s? It wasn’t some neutral scientific consensus - it was a compromise to get generics approved faster so big pharma could keep patent extensions on the brand. The math looks good but the motive? Suspicious. And now they’re using SABE to justify wider ranges for high-variability drugs? That’s just letting them slip in subpar products under the radar. They’re playing us.
Miranda Anderson- 7 March 2026
I read this whole thing and honestly it made me feel a little better about the generic I’ve been taking for my cholesterol. I used to worry every time I got a different bottle - different color, different logo - but now I understand it’s not about how it looks or what company made it. It’s about what your body actually does with the medicine. The part about log-normal distribution and why they use logs blew my mind. I’m not a scientist but I get it now. It’s not magic, it’s math. And the fact that they’ve been testing this for decades on real people… that’s reassuring. I used to think I was being scammed. Now I feel like I’m part of a system that actually works. Thank you for explaining it so clearly.
Gigi Valdez- 8 March 2026
The 80-125% bioequivalence criterion represents a scientifically validated threshold for therapeutic interchangeability. Its adoption by international regulatory agencies reflects consensus derived from extensive pharmacokinetic modeling and clinical outcome data. While public perception often misinterprets the rule as a measure of active pharmaceutical ingredient content, the regulatory framework explicitly distinguishes between chemical composition and pharmacokinetic performance. The use of log-transformation to normalize skewed data and the application of 90% confidence intervals are methodologically sound practices grounded in biostatistical principles. The evidence supporting the safety and efficacy of approved generics remains robust and consistent across population-level studies.
Sneha Mahapatra-10 March 2026
❤️ Thank you for writing this. I’ve been on levothyroxine for 12 years and switched generics twice. I panicked both times. But after reading this, I finally understand why my doctor said ‘it’s fine’ even when the pill looked different. The 90-111% range for narrow drugs makes so much sense now. I didn’t know about SABE or log-normal distributions - but I get that this isn’t about profit. It’s about protecting people. I’m going to stop scrolling through Reddit fear-mongering threads. This was healing. I feel less alone now.
Charity Hanson-10 March 2026
YESSSSSSS this is the kind of info we NEED to spread! So many people are scared of generics because they think they’re ‘fake’ or ‘weak’ - but nope! It’s science! And it’s working! 💪 I work in a pharmacy and I’ve seen patients cry because they can’t afford the brand. Then they try the generic and boom - same results. Same life. Same health. We need more posts like this. Share it. Tag your friends. Let’s stop the stigma! 🌍❤️
Justin Ransburg-11 March 2026
Thank you for this incredibly thorough breakdown. It’s rare to see a post that doesn’t just dismiss concerns but addresses them with evidence. The part about the Complex Generics Initiative and the $35M annual investment was eye-opening. It shows regulators aren’t cutting corners - they’re investing heavily to get it right. This isn’t just about cost savings. It’s about access. And access to life-saving medicine is a human right. This deserves to be shared with every patient who’s ever been told ‘it’s just a generic’ like it’s a downgrade.
Sumit Mohan Saxena-12 March 2026
It is imperative to emphasize that the 80-125% bioequivalence criterion is not a measure of pharmacological potency per se, but rather a quantification of systemic exposure. The geometric mean ratio of AUC(0-t) and Cmax, with its 90% confidence interval constrained within the specified limits, ensures that the rate and extent of absorption are therapeutically equivalent. Furthermore, the utilization of logarithmic transformation is not merely a mathematical convenience but a necessary correction for inherent skewness in pharmacokinetic data. Regulatory bodies have consistently validated this approach through empirical observation over decades. Consequently, the claim that generic medications are inferior is empirically unsupported.
Brandon Vasquez-13 March 2026
My dad’s on warfarin. We switched generics last year. He was terrified. We talked to his pharmacist, read up, and now he’s fine. No INR spikes. No issues. I just wanted to say - this post helped me explain it to him. Not with jargon. Just with facts. That’s all he needed. Thank you.
Byron Duvall-15 March 2026
They say 90% of generics work fine but what about the 10%? Who’s paying for the ER visits? Who’s covering the lawsuits? This rule lets them skip real testing. They just run a few healthy young guys through a lab and call it good. What about people with liver disease? Diabetes? Old people? The rule doesn’t care. It’s designed to make money, not save lives.