Tag: bioequivalence
Patient Support Groups: Sharing Experiences with Generics
Discover how patient support groups bridge the gap between generic drug science and personal trust. Learn the risks, benefits, and tips for finding safe communities.
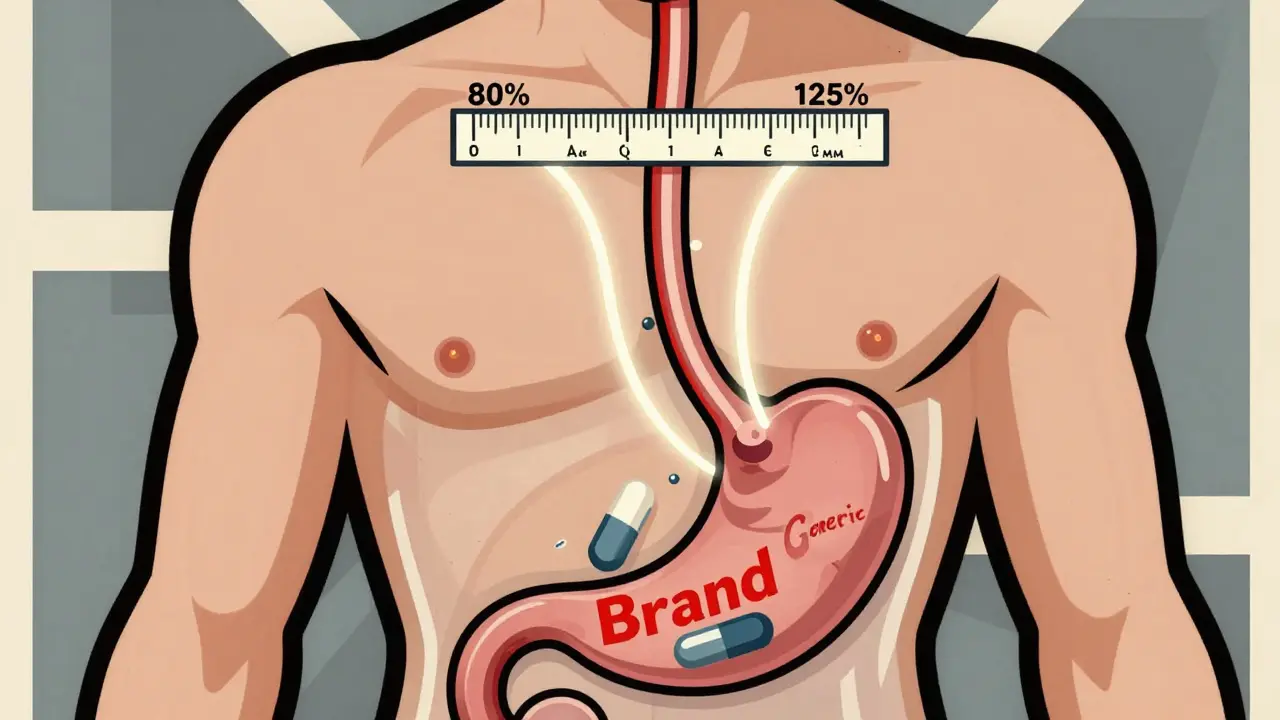
The 80-125% Rule: Understanding Bioequivalence Confidence Intervals for Generic Drugs
The 80-125% rule ensures generic drugs are absorbed the same way as brand-name versions. It's not about drug content-it's about how your body processes it. Here's how it works, why it matters, and what it really means for patients.
Special Populations in Bioequivalence: How Age and Sex Impact Generic Drug Testing
Bioequivalence studies for generic drugs have historically excluded women and older adults. New regulations now require more representative testing to ensure safety and effectiveness across all users.
Drug Interactions: Same Risk for Generic and Brand Medications
Generic and brand-name drugs have the same active ingredients and carry the same risk of drug interactions. The FDA and major studies confirm they're equally safe and effective. Here's what you really need to know.