Author: Caspian Hartwell
Patient Support Groups: Sharing Experiences with Generics
Discover how patient support groups bridge the gap between generic drug science and personal trust. Learn the risks, benefits, and tips for finding safe communities.
L-Tryptophan and Antidepressants: Serotonergic Overlap and Safety
Explore the safety risks of mixing L-Tryptophan supplements with antidepressants. Understand serotonin syndrome, dosage limits, and medical guidelines.
Weight Loss for NAFLD: Diet, Exercise, and Medication Options That Actually Work
Weight loss is the most effective way to reverse fatty liver disease. Learn how diet, exercise, and the new FDA-approved drug semaglutide can help reduce liver fat and prevent serious complications.
Opioid Agreements: Safe Prescribing and Monitoring Practices
Opioid agreements are clinical tools used with PDMPs to ensure safe prescribing and monitor patients on long-term opioid therapy. They reduce misuse, prevent doctor shopping, and improve patient safety when combined with real-time data and EHR integration.
Evergreening: How Pharma Brands Delay Generic Drugs to Protect Profits
Evergreening lets pharmaceutical companies extend patent life on drugs through minor tweaks, delaying cheaper generics and keeping prices high. Learn how it works, why it's controversial, and where it's starting to crack.
Targeted Therapy: How Tumor Genetics Are Changing Cancer Treatment
Targeted therapy uses tumor genetics to attack cancer with precision, avoiding the harsh side effects of chemotherapy. Learn how biomarker testing is changing survival rates - and why access remains unequal.
Hatch-Waxman Act: How It Created the Modern Generic Drug Market in the U.S.
The Hatch-Waxman Act of 1984 created the modern U.S. generic drug market by balancing patent protections for innovators with faster approval for generics. It now governs 90% of prescriptions and saves $313 billion annually.
Polypharmacy in Elderly Patients: How to Manage Multiple Medications Safely
Polypharmacy in elderly patients - taking five or more medications - increases risks of falls, confusion, and hospitalization. Learn how deprescribing, medication reviews, and teamwork between doctors and pharmacists can improve safety and quality of life for seniors.
G6PD Deficiency and Medications: How to Prevent Hemolysis
G6PD deficiency can cause life-threatening hemolysis when exposed to certain medications. Learn which drugs to avoid, safe alternatives, and how testing prevents emergencies. Over 400 million people worldwide are affected - prevention saves lives.
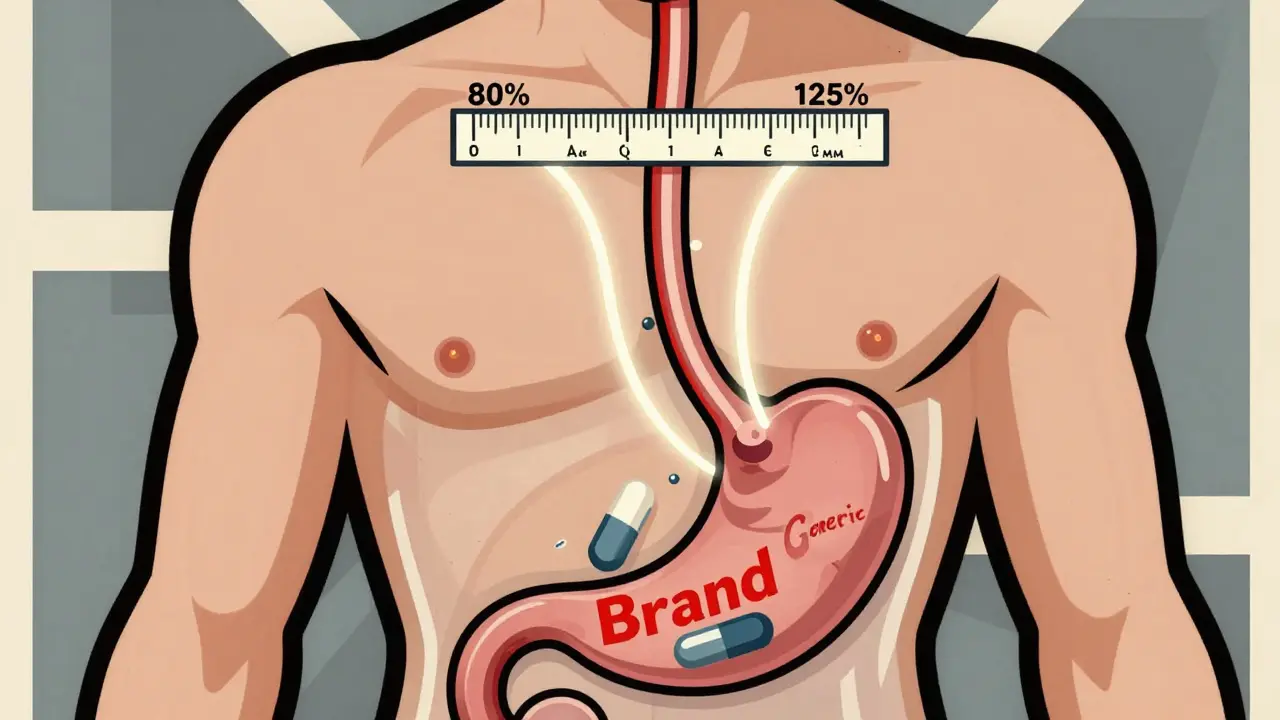
The 80-125% Rule: Understanding Bioequivalence Confidence Intervals for Generic Drugs
The 80-125% rule ensures generic drugs are absorbed the same way as brand-name versions. It's not about drug content-it's about how your body processes it. Here's how it works, why it matters, and what it really means for patients.